The Substance Abuse and Mental Health Services Administration within the Department of Health and Human Services, is asking for input on the Mandatory Guidelines for Federal Workplace Testing Programs and possible updates to the Urine and Oral Fluid Analyte Table to include fentanyl.
Feds Looking to Add Fentanyl to Drug Testing Table
The U.S. Department of Health and Human Services wants public comment on adding fentanyl to the Urine and Oral Fluid Analyte Table, which would affect DOT drug testing for truck drivers and other safety-sensitive workers.

The Substance Abuse and Mental Health Services Administration is considering adding fentanyl to the Urine and Oral Fluid Analyte Table, which could impact workplace drug testing.
Photo: DEA
SAMHSA published a notice in the Federal Register announcing that its Center for Substance Abuse Prevention’s Drug Testing Advisory Board will convene via web conference on Dec. 5, and the board will be discussing adding fentanyl to the analyte table.
The Department of Transportation must follow the HHS scientific guidelines for DOT-regulated drug testing laboratory procedures, so participants in the DOT transportation industry drug testing program should be aware HHS is considering this issue.
Any change to the HHS analyte table may affect the DOT testing program under 49 CFR Part 40, but only after DOT conducts its own rulemaking.
Why HHS Wants to Add Fentanyl to Drug Testing
In its proposal, the agency explained that fentanyl accounts for a large proportion of overdose deaths in the United States and is increasingly used as a stand-alone substance of abuse. According to the National Forensic Laboratory Information System 2021 report, fentanyl was the fourth most frequently identified drug and accounted for nearly 12% of all drugs reported by forensic laboratories.
The Trucking Cares Foundation, the charitable arm of the American Trucking Associations, recently donated $25,000 to Voices for Awareness, a national organization aimed at raising awareness of the dangers of illicit fentanyl in the U.S.
“The epidemic of deadly fentanyl is having a devastating effect on individuals, families, and communities," said TCF Chairman Phil Byrd. "The Trucking Cares Foundation is determined to raise awareness about this growing crisis and recognize elected officials who are shaping our government’s response."
How to Comment
SAMHSA is requesting public comment on the recommendation of adding fentanyl/norfentanyl (along with their proposed testing cutoffs) to its analyte table.
Comments can be submitted prior to the DTAB meeting, during the DTAB meeting, and up to 30 days after the DTAB meeting, but no later than January 4, 2024.
Written comments regarding the addition of fentanyl to the HHS analyte table can be submitted via e-mail to: DFWP@samhsa.hhs.gov.
More Safety & Compliance

'Beyond Compliance,' Regulations, Driver Coaching on ATRI’s 2026 Research List
The American Transportation Research Institute will examine driver coaching, regulatory impacts — including the "Beyond Compliance" concept —and weather disruptions that shape trucking operations.
Read More →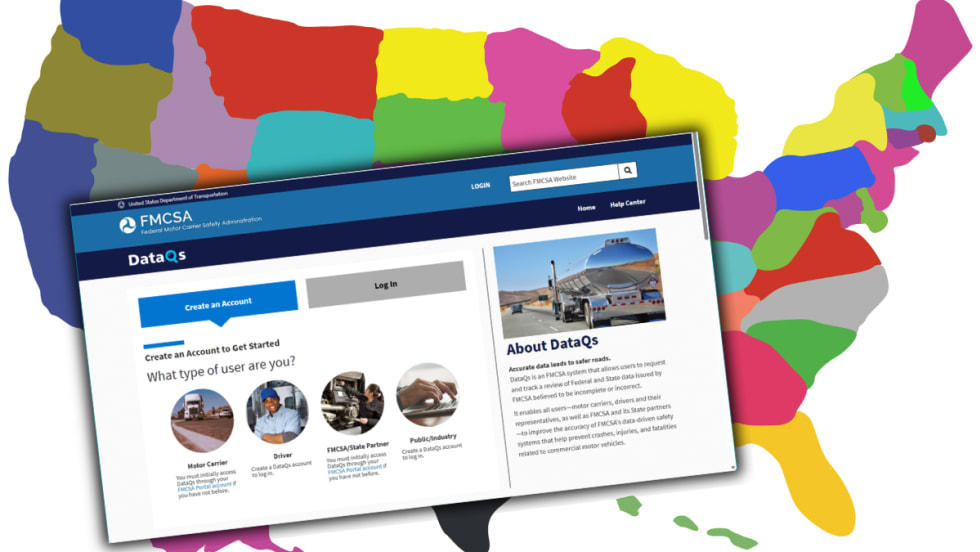
FMCSA Revamps DataQs to Improve Fairness, Speed of Reviews
New requirements add firm deadlines and independent review steps, addressing long-standing complaints about inconsistent rulings and slow response times.
Read More →
FMCSA Extends Paper Medical Card Exemption … Again
Five states still aren't ready to accept commercial driver medical exam information directly from the medical examiner's registry.
Read More →
HDT Honors the Best New Products of 2025 at TMC [Photos]
Heavy Duty Trucking's Top 20 Products awards recognize the best new products and technologies. Check out the award presentations at the 2026 Technology & Maintenance Council annual meeting.
Read More →
Detroit Engines: Trusted Performance, Built for What's Next
The Detroit® Gen 6 engine platform proves that real progress doesn’t require a complete redesign. Built on 20 years of trusted technology, these engines are designed for efficiency, stronger performance, and greater reliability than before. And they do it all while complying with 2027 EPA standards on every mile.
Read More →
Aperia Expands Halo Platform with Steer-Tire Inflation System, Fifth-Wheel Integration
Aperia Technologies introduced a new automatic tire inflation system for steer axles and a partnership with Fontaine Fifth Wheel to integrate coupling status into its Halo Connect platform.
Read More →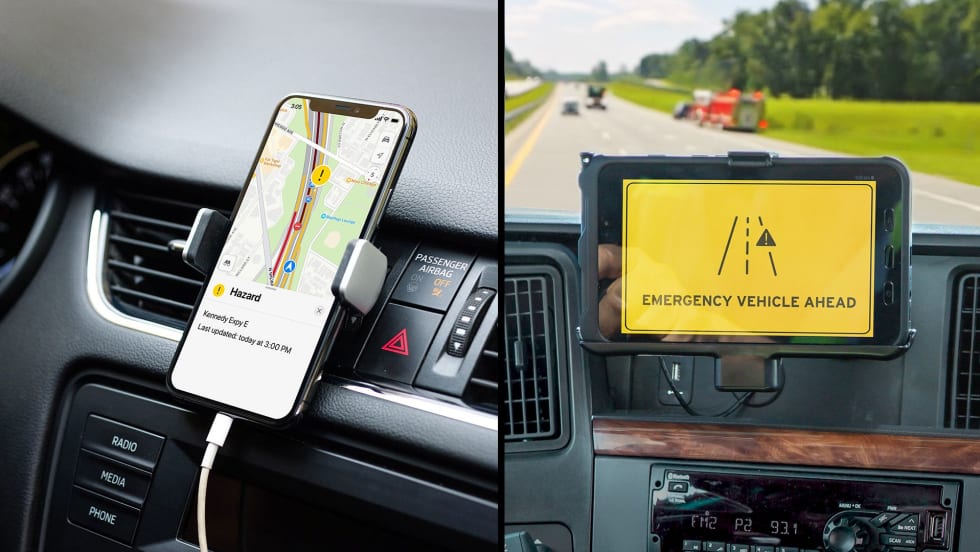
Fleetworthy and HAAS Alert Expand Partnership Stopped Truck Protection Alerts
Fleetworthy and HAAS Alert expanded their partnership to deliver real-time digital alerts that warn motorists when commercial trucks are stopped roadside and notify truck drivers when approaching emergency responders.
Read More →
New Entrants, Chameleon Carriers, and Safety: Is It Too Easy to Start a Trucking Company?
More than 100,000 new trucking companies enter the industry each year, but regulators manage to audit only a fraction of them. That churn creates opportunities for inexperienced startups — and for “chameleon carriers” that shut down after safety violations and reappear under new identities. Read more from Deborah Lockridge in this commentary.
Read More →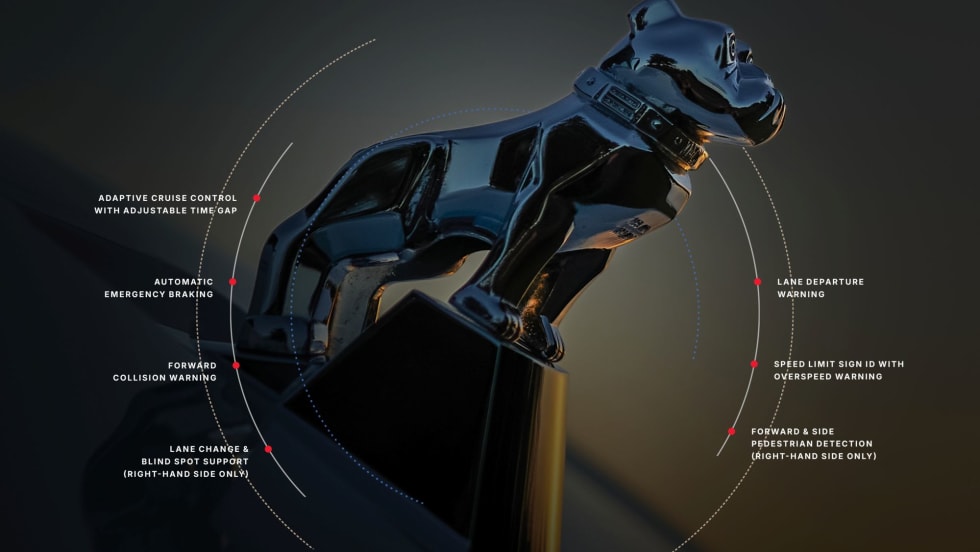
Mack Introduces Mack Protect Collision Mitigation System for MD Series
Mack Trucks has expanded its proprietary Mack Protect collision mitigation platform to the Mack MD Series, bringing heavy-duty safety technology to medium-duty trucks operating in urban and regional environments.
Read More →
Bison Transport, Mill Creek Motor Freight Win TCA Fleet Safety Awards Grand Prize
Two Canadian fleets earned the Grand Prize in the Truckload Carriers Association’s 2025 Fleet Safety Awards, recognizing the industry’s top safety performance based on accident frequency and safety programs.
Read More →


